Our research leverages synthetic biology to develop technologies that overcome challenges related to agriculture, energy, and health. To understand how to design safe biotechnologies for environmental applications, our group is using microbiome-scale information storage to understand the environmental controls on the host range of mobile DNA exchanged via conjugation and phage transduction and to enable the programming of hard-to-manipulate microbes. To generate living electronics for real-time sensing, we are studying sequence-structure-function relationships in a range of proteins, including Fe-S proteins that function as electron transfer hubs, membrane-spanning decaheme cytochromes that mediate extracellular electron transfer, and hemoproteins containing Fe-S clusters that synthesize redox active chemicals. Our ultimate goal is to translate synthetic biology advances into the Earth and environmental sciences. To support these efforts, our research is highly collaborative and frequently involves team projects and training that connect with clinicians, environmental scientists, materials scientists, physicists, public policy researchers, and other synthetic biologists.
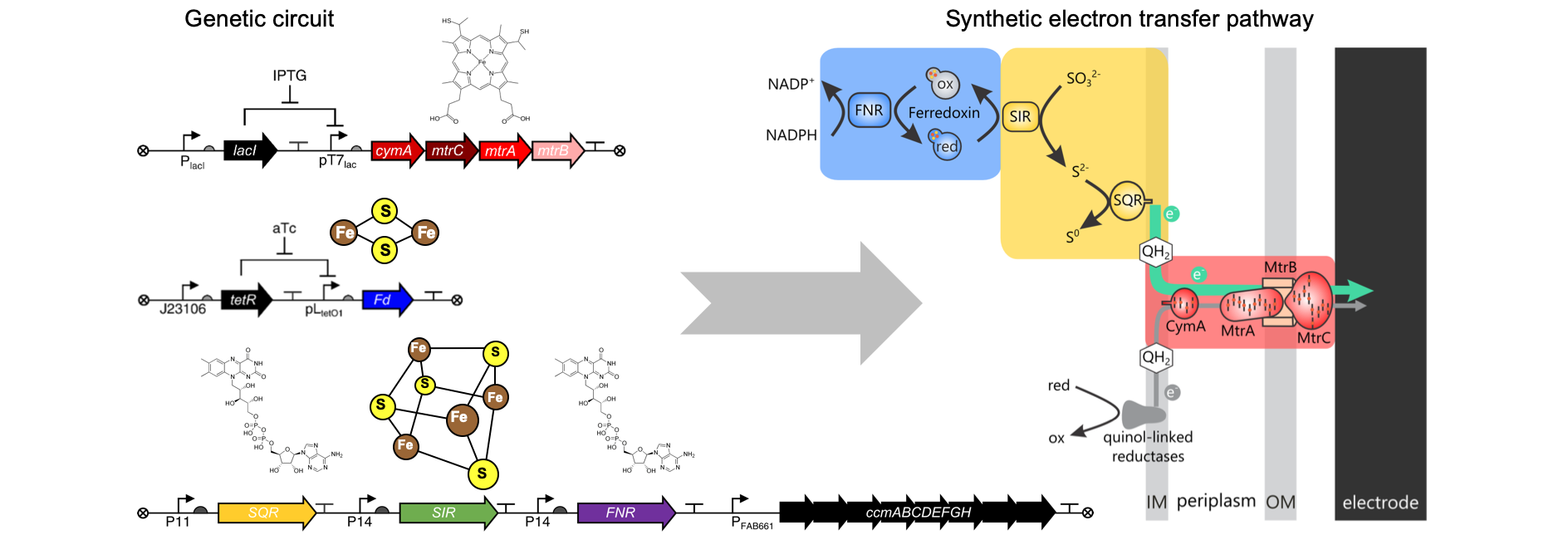
Living electrical sensors. We are building synthetic electron transfer pathways and interfacing cells with materials to create useful bioelectronic technologies. These efforts have yielded the first fast living electronic sensors that convert chemical information in the environment into electrical information at the cell surface in real time (Nature 2022). To support these efforts, we are working to understand how proteins that control extracellular electron transfer can be mutated to create novel components for synthetic biology (Biochemistry 2022). We are also using cellular assays for electron transfer proteins to study how metalloproteins act as electrical hubs to control the proportion of electrons that are transferred between different metabolic pathways in cells (Frontiers Energy Research 2020), to create electrical protein switches using rational design (Nature Chemical Biology 2019) and directed evolution (AIChE Journal 2019), to investigate whether ancient iron-sulfur proteins can support cellular electron transfer (PNAS 2019), and to evaluate how recombination affects electron transfer mediated by metalloproteins (ACS Synthetic Biology 2020).
Environmental synthetic biology. We are working to translate synthetic biology advances for biosensing and information storage into the Earth and environmental sciences by creating novel tools for synthetic biology in soils, sediments, wastewater, and rivers (Frontiers Microbiology 2021). To support this research, we have created a novel family of gas reporting proteins (ACS Synthetic Biology 2020), which allow for nondestructive biosensing within soils using ≤1000 synthetic cells per gram of soil (ACS Synthetic biology 2022). The goal of these studies is to understand how changes in the physical and chemical properties of an environmental niche control plant-microbe signaling (Science Advances 2020), microbe-microbe signaling (ACS Synthetic Biology 2018), and horizontal gene transfer (ES&T 2016). Also, we have been using biosensors in synthetic soils to understand abiotic controls on microbial behaviors (mSystems 2022), such as greenhouse gas production and soil carbon storage. These synthetic materials are useful for studying the mechanisms that underlie environmental controls on microbial fitness, signaling, and gene transfer.
Microbiome engineering. Computers use two mechanisms to store data: read-only memory (ROM), which can be written but not erased, and random-access memory (RAM), which can be written and erased multiple times. Cells can also be programmed to store high density read-only data by permanently modifying the genetic code, and they have the potential to be used as sustainable data storage components for digital devices. However, it remains challenging to store high density data in cells using RAM-like approaches that allow for multiple write-read-erase cycles. In addition, there are limited methods to read out stored biological information non-disruptively, without affecting cell viability. To overcome these challenges, we are working to code information into different biomolecules within cells, including synthetic RNA, which are retained within cells, and redox-active small molecules, which can diffuse in and out of cells. Outside of cells, the mediators can be non-destructively detected using an electrode.